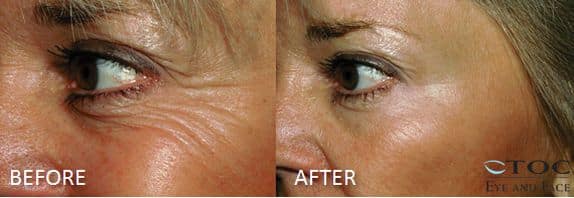
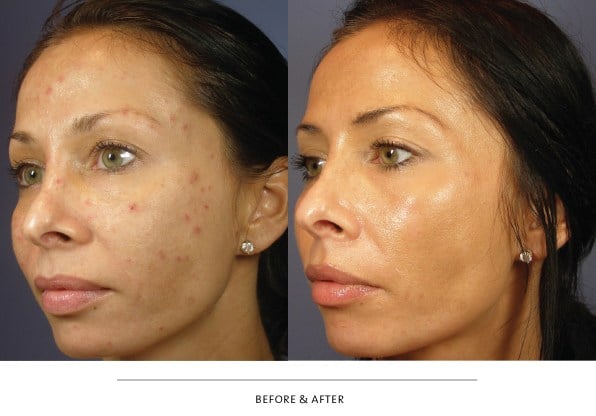
Botox® is one of the most studied, most popular, and possibly the most often prescribed medication in the United States. Most people think of Botox® as a drug used exclusively for treatment of facial wrinkles or to modify facial movement and that it may help them avoid cosmetic surgeries such as, blepharoplasty, forehead lift, or facelift. Botox®, and its cousin, Dysport®, are both effective modulators of muscle action. For this reason, both are useful not only for treatment of facial frown lines, droopy eyebrows (brow ptosis), and crow’s feet wrinkles, but they are also useful in treating other disorders such as hemifacial spasm, blepharospasm, facial tics, and abnormal facial muscle movement following facial trauma or Bell’s Palsy. Doctors and nurses at TOC Eye and Face and TOC Medical Spa have used Botox® and Dysport® to enhance aesthetic results of fillers commonly used in our practice such as, Restylane®, Juvederm®XC, and Radiesse®. We also use it as a supplemental treatment to enhance results following facial laser resurfacing, blepharoplasty and ptosis surgery, endoscopic forehead lifting, and rhinoplasty. Botox® and Dysport® treatments are a powerful adjunct to aesthetic skin care provided by TOC Medical Spa nurses who work hand-in-hand with and under the supervision of TOC physicians. A nice effect of Botox® reduction of wrinkles in the crow’s feet area at the outer corner of the eyelids is evidenced in the before and after treatment photographs above. The relaxing and smoothing effects last between 3 and 4 months. Re-injection is necessary to maintain the relaxing effect.
Botox® and Dysport® are commercial product names for a purified protein produced by a strain of bacteria called Clostridium botulinum. It has been the subject of scientific studies for over 100 years. TOC physicians and nurses have participated in 3 national scientific studies to evaluate the efficacy of the drug for the treatment of facial wrinkles, otherwise known as rhytids. The first study took place in the early to mid-1980’s and resulted in Botox®, known at the time as Oculinum, achieving FDA approval for treatment of strabismus (crossed eyes) and blepharospasm (a disease characterized by severe, uncontrollable eyelid and facial spasms). I was the chief investigator for two other national FDA directed studies conducted to determine the dosage and frequency of administration of newer, sister drugs similar to Botox®. TOC Medical Spa nurse, Andrea Guidry, R.N., was the head nurse involved in one of the studies. She, along with our other five nurses at the TOC Medical Spa, have over 70 years of collective experience in the treatment of facial wrinkles and abnormal sweating (hyperhidrosis) in the axilla (underarm area). These studies, lasting over five years, have given the physicians and nursing staff at TOC Eye and Face and TOC Medical Spa a unique 20-year experience in the use of Botox® and Dysport® in clinical practice. We welcome the opportunity to tell you more and to demonstrate the effect of one of the most popular facial aesthetic treatments in the world.
John W. Shore, M.D.